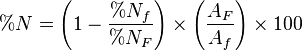Diatomaceous earth (pron.: /ˌdaɪ.ətəˌmeɪʃəs ˈɜrθ/) also known as D.E., diatomite, or kieselgur/kieselguhr, is a naturally occurring, soft, siliceous sedimentary rock that is easily crumbled into a fine white to off-white powder. It has a particle size ranging from less than 3 micrometre to more than 1 millimeter, but typically 10 to 200 micrometres. Depending on the granularity, this powder can have an abrasive feel, similar to pumice powder, and is very light as a result of its high porosity. The typical chemical composition of oven-dried diatomaceous earth is 80 to 90% silica, with 2 to 4% alumina (attributed mostly to clay minerals) and 0.5 to 2% iron oxide.[1]
Diatomaceous earth consists of fossilized remains of diatoms, a type of hard-shelled algae. It is used as a filtration aid, mild abrasive in products including toothpaste, mechanical insecticide, absorbent for liquids, matting agent for coatings, reinforcing filler in plastics and rubber, anti-block in plastic films, porous support for chemical catalysts, cat litter, activator in blood clotting studies, a stabilizing component of dynamite, and a thermal insulator.
Contents |
Geology and occurrence
Diatomaceous earth as viewed under bright field illumination on a light microscope. Diatomaceous earth is made up of the cell walls/shells of single cell diatoms and readily crumbles to a fine powder. Diatom cell walls are made up of biogenic silica; silica synthesised in the diatom cell by the polymerisation of silicic acid. This image of diatomaceous earth particles in water is at a scale of 6.236 pixels/μm, the entire image covers a region of approximately 1.13 by 0.69 mm.
Formation
Diatomite forms by the accumulation of the amorphous silica (opal, SiO2·nH2O) remains of dead diatoms (microscopic single-celled algae) in lacustrine or marine sediments. The fossil remains consist of a pair of symmetrical shells or frustules.[1]
Discovery
In 1836 or 1837, the peasant and goods waggoner, Peter Kasten,[2] discovered diatomaceous earth (German: kieselgur) when sinking a well on the northern slopes of the Haußelberg hill, in the Lüneburg Heath in north Germany. Initially, it was thought that limestone had been found, which could be used as fertilizer. Alfred Nobel used the properties of diatomaceous earth in the manufacture of dynamite. The Celle engineer, Wilhelm Berkefeld, recognized its ability to filter, and developed tubular filters (known as filter candles) fired from diatomaceous earth.[3] During the cholera epidemic in Hamburg in 1892, these Berkefeld filters were used successfully.
Extraction and storage sites in the Lüneburg Heath
- Neuohe – extraction from 1863 to 1994
- Wiechel from 1871 to 1978
- Hützel from 1876 to 1969
- Hösseringen from ca. 1880 to 1894
- Hammerstorf from ca. 1880 to 1920
- Oberohe from 1884 to 1970
- Schmarbeck from 1896 to ca. 1925
- Steinbeck from 1897 to 1928
- Breloh from 1907 to 1975
- Schwindebeck from 1913 to 1975
- Hetendorf from 1970 to 1994
The deposits are up to 28 metres (92 ft) thick and are all of freshwater diatomaceous earth.
Until the First World War almost the entire worldwide production of diatomaceous earth was from this region.
Other deposits
In Germany diatomaceous earth was also extracted at Altenschlirf [4] on the Vogelsberg (Upper Hesse) and at Klieken [5] (Saxony-Anhalt).
There is a layer of diatomaceous earth up to 4 metres (13 ft) thick in the nature reserve of Soos in the Czech Republic.
In Colorado and in Clark, Nevada (USA), there are deposits that are up to several hundred metres thick in places.
Sometimes diatomaceous earth is found on the surface in deserts. Research has shown that the erosion of diatomaceous earth in such areas (such as the Bodélé Depression in the Sahara) is one of the most important sources of climate-affecting dust in the atmosphere.
The commercial deposits of diatomite are restricted to Tertiary or Quaternary periods. Older deposits from as early as the Cretaceous Period are known, but are of low quality.[6] Marine deposits have been worked in the Sisquoc Formation in Santa Barbara County, California near Lompoc and along the Southern California coast. Additional marine deposits have been worked in Maryland, Virginia, Algeria and the MoClay of Denmark. Fresh water lake deposits occur in Nevada, Oregon, Washington and California. Lake deposits also occur in interglacial lakes in the eastern US and Canada and in Europe in Germany, France, Denmark and the Czech Republic. The worldwide association of diatomite deposits and volcanic deposits suggests that the availability of silica from volcanic ash may be necessary for thick diatomite deposits.[6]
Applications
Industrial
In 1866, Alfred Nobel discovered that nitroglycerin could be made much more stable if absorbed in diatomite. This allows much safer transport and handling than nitroglycerin in its raw form. He patented this mixture as dynamite in 1867, and the mixture is also referred to as guhr dynamite.
Filtration
One form of diatomaceous earth is used as a filter medium, especially for swimming pools. It has a high porosity, because it is composed of microscopically small, coffin-like, hollow particles. Diatomaceous earth (sometimes referred to by trademarked brand names such as Celite) is used in chemistry as a filtration aid, to filter very fine particles that would otherwise pass through or clog filter paper. It is also used to filter water, particularly in the drinking water treatment process and in fish tanks, and other liquids, such as beer and wine. It can also filter syrups, sugar, and honey without removing or altering their color, taste, or nutritional properties.[7]
Abrasive
The oldest use of diatomite is as a very mild abrasive and, for this purpose, it has been used both in toothpaste and in metal polishes, as well as in some facial scrubs.
Pest control
Diatomite is used as an insecticide, due to its abrasive and physico-sorptive properties.[8] The fine powder absorbs lipids from the waxy outer layer of insects’ exoskeletons, causing them to dehydrate.[9] Arthropods die as a result of the water pressure deficiency, based on Fick’s law of diffusion. This also works against gastropods and is commonly employed in gardening to defeat slugs. However, since slugs inhabit humid environments, efficacy is very low. It is sometimes mixed with an attractant or other additives to increase its effectiveness. Medical-grade diatomite is sometimes used to de-worm both animals and humans, with questionable efficacy.[10][11] It is commonly used in lieu of boric acid, and can be used to help control and possibly eliminate bed bug, house dust mite, cockroach and flea infestations.[citation needed][12][13] This material has wide application for insect control in grain storage.[14]
In order to be effective as an insecticide, diatomaceous earth must be uncalcinated (i.e., it must not be heat-treated prior to application)[15] and have a mean particle size below about 12 microns (i.e., food-grade— see below).
Although considered to be relatively low-risk, pesticides containing diatomaceous earth are not exempt from regulation in the United States under the Federal Insecticide, Fungicide, and Rodenticide Act and must be registered with the Environmental Protection Agency.[16]
Absorbent
Its absorbent qualities make it useful for spill clean-up and the U.S. Centers for Disease Control recommends it to clean up toxic liquid spills. These qualities also lend themselves to use in facial masks to absorb excess oils.
It has been employed as a primary ingredient in a type of cat litter. The type of silica used in cat litter comes from freshwater sources and does not pose a significant health risk to pets or humans.
The microscopic matrix of DE make it a highly effective desiccant; it can absorb up to six times its weight in water.[citation needed] It also can be used in the absorption of Ethylene Gas to reduce decay in fruits and produce.[citation needed] The combination of refrigeration and DE as a filter medium is one of the best ways to extend shelf life of fruits and produce commercially and in a home refrigerator.[citation needed] One should use a food grade, as opposed to pool grade, DE when using it as a desiccant filter near fruit & produce.
Thermal
Its thermal properties enable it to be used as the barrier material in some fire resistant safes.[citation needed] It is also used in evacuated powder insulation for use with cryogenics.[17] Diatomaceous earth powder is inserted into the vacuum space to aid in the effectiveness of vacuum insulation. It was used in the Classical AGA Cookers as a thermal heat barrier.
DNA purification
Diatomite (Celite) can be used for the removal of DNA in the presence of a highly concentrated chaotropic agent such as sodium iodide, guanidinium chloride and guanidinium thiocyanate.[citation needed] As with other silicates, the diatomites will remove double stranded DNA but not RNA or proteins. The DNA can be extracted from the diatomites using low ionic strength buffers, including water, at neutral to slightly alkaline pH.[citation needed] Crude diatomites of a uniform size must first be washed in a heated acid such as 5M HCl.[18] Calcination can further improve consistency of the material, while mild caustic treatment may improve adsorption with lower levels of chaotrophs.[citation needed]
Use in agriculture
Natural freshwater diatomaceous earth is used in agriculture for grain storage as an anticaking agent, as well as an insecticide.[19] It is approved by the US Department of Agriculture as a feed supplement to prevent caking.(http://www.ars.usda.gov/is/np/mba/apr00/food.htm)
It is also used as a natural anthelmintic (dewormer). Some farmers add it to their livestock and poultry feed to prevent the caking of feed.[20] “Food Grade Diatomaceous Earth” is widely available in agricultural feed supply stores. DE is acceptable as an anti caking agent for livestock.(http://www.ars.usda.gov/is/np/mba/apr00/food.htm)
Hydroponics
Freshwater diatomite can be used as a growing medium in hydroponic gardens.
It is also used as a growing medium in potted plants, particularly as bonsai soil. Bonsai enthusiasts use it as a soil additive, or pot a bonsai tree in 100% diatomaceous earth. Like perlite, vermiculite, and expanded clay, it retains water and nutrients, while draining fast and freely, allowing high oxygen circulation within the growing medium.
Marker in livestock nutrition experiments
Natural diatomaceous earth (dried, not calcined) is regularly used in livestock nutrition research as a source of acid insoluble ash (AIA), which is used as an indigestible marker. By measuring the content of AIA relative to nutrients in test diets and feces or digesta sampled from the terminal ileum (last third of the small intestine) the percentage of that nutrient digested can be calculated using the following equation:
- Where:
 is percent Nutrient Digestibility
is percent Nutrient Digestibility is the percent of nutrients in the feces
is the percent of nutrients in the feces is the percent of nutrients in the feed
is the percent of nutrients in the feed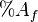 is the percent of AIA in the feces
is the percent of AIA in the feces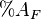 is the percent of AIA in the feed
is the percent of AIA in the feed
- And:
Natural diatomaceous earth (freshwater) is preferred by many researchers over chromic oxide, which has been widely used for the same purpose, but which is also a known carcinogen and therefore a potential hazard to research personnel.
Construction
The spent diatomaceous from the brewing process can be mixed with the ceramic mass for the production of red bricks.[21]
Specific varieties
- Tripolite is the variety found in Tripoli, Libya.
- Bann clay is the variety found in the Lower Bann valley in Northern Ireland.
- Moler (Mo-clay) is the variety found in northwestern Denmark, especially on the islands of Fur and Mors.
- Freshwater-derived food grade diatomaceous earth is the type used in US agriculture for grain storage, as feed supplement, and as an insecticide. It is produced uncalcinated, has a very fine particle size, and is very low in crystal silica (<2%).
- Salt-water-derived pool/ beer/ wine filter grade is not suitable for human consumption nor is effective as an insecticide. It is usually calcinated before being sold to remove impurities and undesirable volatile contents, is composed of larger particles than the freshwater version, and has a high silica content (>60%).
Climatologic importance
The Earth’s climate is affected by dust in the atmosphere, so locating major sources of atmospheric dust is important for climatology. Recent research indicates that surface deposits of diatomaceous earth play an important role. For instance, the largest single atmospheric dust source is the Bodélé depression in Chad, where storms push diatomite gravel over dunes, generating dust by abrasion.[22]
Safety considerations
Today’s common DE formulations are safe to use as they are predominantly made up of amorphous silica and contain little or no crystalline silica. The crystalline silica content of DE is regulated in the United States by the Occupational Safety and Health Administration (OSHA), and there are guidelines for the maximum amounts allowable in the product and in the air near the breathing zone of workers.[23]
In the 1930’s, long term occupational exposure among workers in the cristobalite DE industry who were exposed to high levels of airborne crystalline silica over decades were found to have an increased risk of silicosis.[24] Today, workers are required to use respiratory-protection measures when concentrations of silica exceed allowable levels.
Diatomite produced for pool filters is treated with high heat (calcination) and a fluxing agent (soda ash), causing the formerly harmless silicon dioxide to assume its crystalline form.[23]
The age and shape of diatoms
Each deposit of diatomaceous earth is different, with varying blends of pure diatomaceous earth combined with other natural clays and minerals.
The diatoms in each deposit contain different amounts of silica, depending on the age of the deposit. As well, the species of diatom may also differ among deposits.
The species of diatom is dependent upon the age and paleo-environment of the deposit. In turn, the shape of a diatom is determined by its species.
The shape of the diatoms contained in a deposit has not been proven to affect their functionality when it comes to the absorption of liquids, however certain applications, such as that for slugs and snails, do work best when a particular shaped diatom is used. For example, in the case of slugs and snails large, spiny diatoms work best to lacerate the epithelium of the mollusk. Diatom shells will work to some degree on vast majority of animals that undergo ecdysis in shedding cuticle, such as arthropods or nematodes. It may have some effect also on lophotrochozoans, such as mollusks or annelids.
Many deposits throughout British Columbia, Canada, such as Red Lake Earth, are from the Miocene age and contain a species of diatom known as Melosira granulate. These diatoms are approximately 12 to 13 million years old and are a small globular shape.
A deposit containing diatoms from this age can provide many more benefits than that of an older deposit. For example, diatoms from the Eocene age (approximately 40 to 50 million years old) are not as effective in their ability to absorb fluids because older diatoms such as this recrystallize, their small pores becoming filled with silica.[25]
See also
References
- ^ a b Antonides, Lloyd E. (1997). Diatomite (PDF). U.S.G.S.. Retrieved 2010-12-12.
- ^ http://www.humboldt-foundation.de/kosmos/kultur/2001_002.htm + Heinrich Küsel „Der Speicher“ v.1930[dead link]
- ^ ELGA Berkefeld Water Treatment (Wasseraufbereitung)
- ^ http://www2.natpa.de/bonifatius/senken/p7.htm Über den früheren Abbau von Kieselgur im Vogelsberg/Hessen
- ^ http://homepages.compuserve.de/tmby100/kieselgur.htm Geschichte des Kieselgurabbaus in Klieken
- ^ a b Cummins, Arthur B., Diatomite, in Industrial Minerals and Rocks, 3rd ed. 1960, American Institute of Mining, Metallurgical, and Petroleum Engineers, pp. 303 – 319
- ^ Root, A.I.; E.R. Root (March 1, 2005). The ABC and xyz of bee culture. Kessinger Publishing. p. 387. ISBN 978-1-4326-2685-3. Retrieved March 8, 2011
- ^ Fields, Paul; Allen, Sylvia; Korunic, Zlatko; McLaughlin, Alan; Stathers, Tanya (July 2002). “Standardized testing for diatomaceous earth”. Proceedings of the Eighth International Working Conference of Stored-Product Protection. York, U.K.: Entomological Society of Manitoba. http://home.cc.umanitoba.ca/~fieldspg/fields/de-test-p.pdf.
- ^ Staff. “Natural Methods for Controlling Fleas.”http://www.xmission.com/~emailbox/fleas.htm
- ^ Lartigue, E. del C.; Rossanigo, C. E. (2004). “Insecticide and anthelmintic assessment of diatomaceous earth in cattle”. Veterinaria Argentina 21 (209): 660–674.
- ^ Fernandez, M. I.; Woodward, B. W.; Stromberg, B. E. (1998). “Effect of diatomaceous earth as an anthelmintic treatment on internal parasites and feedlot performance of beef steers”. Animal Science 66 (3): 635–641. doi:10.1017/S1357729800009206.
- ^ http://www.springerlink.com/content/a3u1r27425138354/
- ^ http://cat.inist.fr/?aModele=afficheN&cpsidt=17999828
- ^ http://www.survival-center.com/foodfaq/ff17-oxy.htm Survival Information Center
- ^ Capinera, John L. (2008). “Diatomaceous earth”. In Capinera, John L. Encyclopedia of Entomology (Second ed.). Springer. p. 1216. ISBN 9781402062421.
- ^ http://www.epa.gov/pesticides/regulating/labels/labels_faq/lr_faq_1.html
- ^ Flynn, Thomas M. “Cryogenic Equipment and Cryogenic Systems Analysis.” Cryogenic Engineering. Boca Raton [etc.: CRC, 2005. Print.
- ^ Goren, R.; Baykara, T.; Marsoglu, M. (2002). “A study on the purification of diatomite in hydrochloric acid”. Scand. J. of Metallurgy 31 (2): 115–119. doi:10.1034/j.1600-0692.2002.310205.x.
- ^ Manitoba Agriculture, Food and Rural Initiatives
- ^ (Diatomaceous Earth (DE)
- ^ Ferraz et al., (2011), Manufacture of ceramic bricks using recycled brewing spent kieselguhr. Materials and Manufacturing Processes, 26 (10), 1319-1329. DOI: 10.1080/10426914.2011.551908
- ^ Washington et al., Geophys. Res. Lett. 33 (2006) L09401 doi:10.1029/2006GL025827.
- ^ a b Inert Dusts at Kansas State University
- ^ Radiographic Evidence of Silicosis Risk in the Diatomaceous Earth Industry
- ^ “Diatoms”. UCL London’s Global University. Retrieved September 14, 2011.
External links
- International Chemical Safety Card 0248
- Diatomite: Statistics and Information – USGS
- Tripolite: Tripolite mineral data Citat: “…A diatomaceous earth consisting of opaline silica…”
- DIATOMACEOUS EARTH: A Non Toxic Pesticide
This article uses material from the Wikipedia article Diatomite, which is released under the Creative Commons Attribution-Share-Alike License 3.0.